classwork - Chemistry
Periodic Table of Elements - The Quest
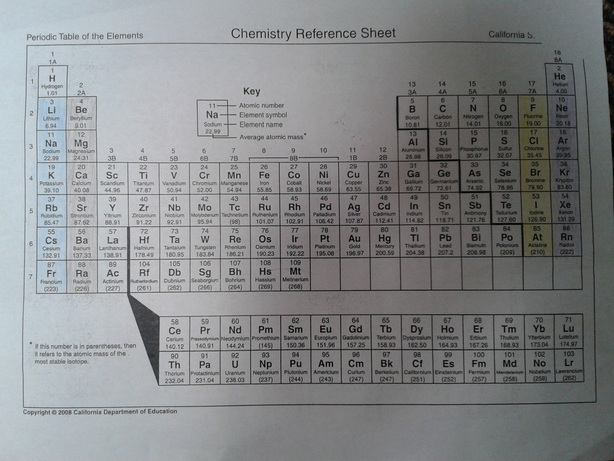
For this in-class mini project we had to research the different sections of the periodic table, Metals, Non metal, Noble Gases, Alkali metals ETC. The blue in the image is Alkali metals, the brown is Alkali earth metals, the yellow is Halogens and finally orange the noble gases. The black line near the top right of the image separates Metals from Non metals.
Fun Facts:
Alkali metals can combust if they are in water.
Alkali earth metals are very reactive and not found in nature.
Halogens all have 7 electrons in their outer shell.
Noble gases have the maximum number of electrons in their outer shell. The maximum being 8.
Fun Facts:
Alkali metals can combust if they are in water.
Alkali earth metals are very reactive and not found in nature.
Halogens all have 7 electrons in their outer shell.
Noble gases have the maximum number of electrons in their outer shell. The maximum being 8.
Summaries of the periodic table
Significant things
There are many important and interesting things about the periodic table. One of the most important things to note is that the number of positive protons determines the atomic number, therefore an atom can not get or lose positive protons without becoming a different element. Electron behavior drives the periodic table. Other things to note is that the first row is all the Alkali metals the second row is all the Alkali earth metals. On the far right row 17, are the halogens and the 18th row are the noble gases. The table has eighteen columns and seven rows with an extra "Landing strip" at the bottom.
Evidence
There are many people who helped and contributed to the periodic table to make it what it is today. The periodic table was "invented" in 1869 by Dmitri Mendeleev. Gilbert lewis was another important figure in the history of the table. His electron work was very important for acids and bases. Plato the greek philosopher minted the word "Elements" as a general term for different small particles of matter.
Significant things
There are many important and interesting things about the periodic table. One of the most important things to note is that the number of positive protons determines the atomic number, therefore an atom can not get or lose positive protons without becoming a different element. Electron behavior drives the periodic table. Other things to note is that the first row is all the Alkali metals the second row is all the Alkali earth metals. On the far right row 17, are the halogens and the 18th row are the noble gases. The table has eighteen columns and seven rows with an extra "Landing strip" at the bottom.
Evidence
There are many people who helped and contributed to the periodic table to make it what it is today. The periodic table was "invented" in 1869 by Dmitri Mendeleev. Gilbert lewis was another important figure in the history of the table. His electron work was very important for acids and bases. Plato the greek philosopher minted the word "Elements" as a general term for different small particles of matter.